
Isotopic analysis has numerous applications within the discipline of archaeology, anthropology, and geoarchaeology. In particular, uranium-thorium, lead, and strontium isotope testing can be used to research such topics as migration studies, paleoclimatology, environmental reconstruction, anthropology, and geochronology.
Strontium Isotopes Tell Us Where Our Ancestors Came From
Strontium (Sr) isotope ratio data can be used to reconstruct migration through time. Strontium has four naturally occurring stable isotopes: 84Sr, 86Sr, 87Sr, and 88Sr. 87Sr is radiogenic with a half-life of 48.8 billion years, resulting from the β-decay of 87Rb (rubidium). Approximately 83% of naturally occurring strontium is 88Sr. When answering research questions about migration and mobility studies, the 87Sr/86Sr ratio is applicable due to their similar abundances (7% and 10%), high mass and distinct natural spatial variability.1
Trace amounts of strontium are present in rocks. The isotopic signature is transferred to overlaying soil and water – ultimately taken in by plants and animals as they consume the water and plant matter. As rock weathers and traverses the food chain, the Sr ratio remains fairly constant due to its high mass, making it possible to connect a sample to a specific location. 87Sr/86Sr analysis of migration is applicable to sample types including bone, hair, and tooth enamel.
The geochemical origin of a sample is represented by the ratio of 87Sr/86Sr. A unique
Strontium Isotopes For Tracking Human Mobility: A Case Study
In a 2016 study by Wang et al., the authors used strontium isotope analysis to trace mobility within the early Iron Age populations of the Pamir Plateau in Eurasia. There has been some evidence that inner-Asian populations partook in long-distance trade and migration with other pre-Silk Road cultures with the discovery of foreign material culture such as silk thread.2 These hypotheses were strengthened via strontium isotope research of human remains from a 2,500 year old cemetery on the Pamir Plateau. Isotopic variability in 87Sr/86Sr ratios in dental enamel samples taken from 34 sets of human remains suggest the presence of a highly mobile population. The majority of the remains (24 individuals) were shown to be local to the area, whilst 10 of the individuals possessed ratios suggesting they were migrants.3 This study demonstrates the great potential for understanding human migration patterns, mobility, and trade associations utilizing 87Sr/86Sr ratios.
Developing Uranium-Thorium Chronologies to Reconstruct Past Environments
Uranium-Thorium (U-Th) dating is used to estimate sample ages and develop chronologies for paleoclimate research and environmental reconstruction. While radiocarbon dating measures the quantity of decay of the carbon-14 isotope (14C), U-Th dating measures a decay chain ratio between 234U/230Th. This measurement allows the time that has passed since the sample was formed to be calculated, resulting in the age of the sample.
At the time of formation, the U-Th ratio is initially completely composed of uranium. Over time, the uranium decays into thorium, giving the sample a higher concentration of thorium. Analysis is based on a ratio of parent (234U) and daughter (230Th) atoms in a given sample and measures their isotope activity, calculating the decay of the parent and production of the daughter over time. Younger samples will have had less time to decay, giving them a higher proportion of parent atoms while older samples will have more daughter atoms. U-Th chronology development is based on a couple of assumptions. First, it is assumed that the sample is behaving as a close system with respect to uranium and thorium, meaning there is no elemental exchange between the sample and surrounding environment. Samples which are susceptible to such exchanges (e.g. bone, foraminifera, shells) are not suitable for this method. To be able to calculate the age, the initial thorium value in the sample (Th0) at the time of formation must be known. Since the Th0 in the sample is not known, it is assumed that the sample contains the average crustal detrital thorium value at the time of formation.
__________________________________
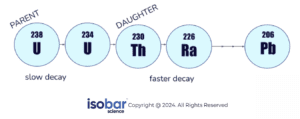
The uranium decay series
__________________________________
The ratio between the parent and daughter atoms allows the sample age to be determined. U-Th dating has a dating limit of up to 500,000 years, making it a better option than other methods with lower dating thresholds such as radiocarbon dating for certain sample types.4 U-Th dating is applicable to calcium carbonate (CaCO3) bearing samples including cave art, speleothems and flowstones, and coral. It is important to be aware of whether a sample comes from an open system or a closed system as it impacts the accuracy of the dating results. The U-Th dating method is based on the assumption that the material is a closed system (e.g. corals, speleothems), meaning that the material hasn’t exchanged uranium or thorium with the surrounding environment. On the other hand, open system samples (e.g. bone, sediments) allow for the exchange of matter (including uranium and thorium), producing lower certainty results. Therefore, the most optimal samples for U-Th dating are those that represent closed systems.5
Lead Isotopes Reveal Human Environmental Impact And Metal Artifact Provenance
Lead (Pb) isotope ratios that are within the uranium decay chain are a function of the amount of uranium and thorium present. The amount of U and Th present is affected by geological processes. For this reason, lead isotopes are a useful tool for answering research questions about the nature and timing of these geological processes.6 The lead isotopic composition of geologic material is a function of three independent decay chains, contributing to potential isotopic variability in minerals. Pb measurement is applicable to igneous and metamorphic rocks, mineral dust, marine and lacustrine sediments, bones, tooth enamel, soil, and others. Testing has a variety of applications including geochemical fingerprinting, and contaminant source tracing.
Geochemical fingerprinting uses the unique lead isotopic signature present in different rock lithologies as well as overlying soils to allow specific areas on the earth’s surface to be distinguished. Distinct isotopic ratios are presented by different rocks, a property that is used extensively for provenance studies such as establishing the geological origin of metals. Lead isotopes do not change form during the conversion process from ore to artifact (e.g. lead pigments, coins). Therefore, analysis can shed light on research questions concerned with trade and the geographical movement of objects because information about where the ore was mined is retained by the material. For example, Cooper, H.K. and Simonetti, A. describe the validity of lead isotope analysis for researching archaeometallurgical provenance in their 2021 study.7 They determined the lead isotopic ratios of the native copper deposits in the Arctic and Subarctic regions of northwestern North America could be distinguished enough to identify regional differences in their isotopic signatures. The ability to distinguish these signatures could be used to reveal copper trade connections.
Another application of lead isotope measurement, contaminant source tracing, is based on the impact caused by human influences on lead’s biogeochemical cycle. Lead enters the environment via various human activities including mining and smelting. Different lead isotope ranges can be correlated to specific anthropogenic activities and used to measure human impacts on the environment over time. Lead can also be traced in bone and teeth. A 2019 study by Scott et al. measured a toxic level of lead contamination in Roman bones from Londinium.8 These results suggest the potential prevalence of widespread lead contamination in urban Roman settlements. Although the study doesn’t indicate a specific singular source or rule out the potential for post-mortem contamination, it highlights contaminant source tracing as a tool for measuring lead exposure within populations.
Conclusion
Strontium, uranium-thorium, and lead isotope analyses are valuable techniques with many applications for archaeological research. One of these applications uses strontium isotopic analysis to reveal information that is beneficial for investigating human and animal migration and mobility. U-Th dating can be used to obtain chronological data for reconstructing climate and environmental changes through time. Lead isotopes are applicable to research into the geological origin of metal artifacts via geochemical fingerprinting and identifying human activities using contaminant source tracing.
_______________________________
Published with permission of Isobar Science, www.isobarscience.com
Cover image, top left: chenspec, Pixabay
_______________________________
Sources
1: https://isobarscience.com/strontium/application/ Accessed September 27, 2021
2, 3: Wang, X., Tang, Z., Wu, J. et al. Strontium isotope evidence for a highly mobile population on the Pamir Plateau 2500 years ago. Sci Rep 6, 35162 (2016). https://doi.org/10.1038/srep35162 Accessed May 6, 2022.
4: https://isobarscience.com/u-th/application/ Accessed September 27, 2021
5: https://isobarscience.com/u-th-dating-assumptions/ Accessed May 20, 2024.
6: https://isobarscience.com/lead-isotopes/application/ Accessed April 21, 2022
7: Cooper HK, Simonetti A. Lead Isotope Analysis of Geological Native Copper: Implications for Archaeological Provenance Research in the North American Arctic and Subarctic. Minerals. 2021; 11(7):667. https://www.mdpi.com/2075-163X/11/7/667. Accessed May 20, 2024
8: Scott SR, Shafer MM, Smith KE, Overdier JT, Cunliffe B, Stafford TW Jr, Farrell PM (2020) Elevated exposure in Roman occupants of Londinium: new evidence from the archaeological record. Archaeometry 62:109–129. https://onlinelibrary.wiley.com/doi/10.1111/arcm.12513 Accessed May 20, 2024
________________________________
Advertisement




