ARIZONA STATE UNIVERSITY—Scientists have long been able to measure and analyze the fossil skulls of our ancient ancestors to estimate brain volume and growth. The question of how these ancient brains compare to modern human brains and the brains of our closest primate cousin, the chimpanzee, continues to be a major target of investigation.
A new study published in Science Advances used CT-scanning technology to view three-million-year old brain imprints inside fossil skulls of the species Australopithecus afarensis (famous for “Lucy” and “Selam” from Ethiopia’s Afar region) to shed new light on the evolution of brain organization and growth. The research reveals that while Lucy’s species had an ape-like brain structure, the brain took longer to reach adult size, suggesting that infants may have had a longer dependence on caregivers, a human-like trait.
The CT-scanning enabled the researchers to get at two long-standing questions that could not be answered by visual observation and measurement alone: Is there evidence for human-like brain reorganization in Australopithecus afarensis, and was the pattern of brain growth in this species more similar to that of chimpanzees or that of humans?
To study brain growth and organization in A. afarensis, the researchers, including ASU paleoanthropologist William Kimbel, scanned eight fossil crania from the Ethiopian sites of Dikika and Hadar using high-resolution conventional and synchrotron-computed tomography. Kimbel, leader of the field work at Hadar, is director of the Institute of Human Origins and Virginia M. Ullman Professor of Natural History and the Environment in the School of Human Evolution and Social Change.
Lucy’s species inhabited eastern Africa more than three million years ago–“Lucy” herself is estimated to be 3.2 million years old–and occupies a key position in the hominin family tree, as it is widely accepted to be ancestral to all later hominins, including the lineage leading to modern humans.
“Lucy and her kin provide important evidence about early hominin behavior–they walked upright, had brains that were around 20 percent larger than those of chimpanzees, and may have used sharp stone tools,” explains coauthor Zeresenay Alemseged (University of Chicago), who directs the Dikika field project in Ethiopia and is an International Research Affiliate with the Institute of Human Origins.
Brains do not fossilize, but as the brain grows and expands before and after birth, the tissues surrounding its outer layer leave an imprint on the inside of the bony braincase. The brains of modern humans are not only much larger than those of our closest living ape relatives but are also organized differently and take longer to grow and mature. Compared with chimpanzees, modern human infants learn longer and are entirely dependent on parental care for longer periods of time. Together, these characteristics are important for human cognition and social behavior, but their evolutionary origins remain unclear.
The CT scans resulted in high-resolution digital “endocasts” of the interior of the skulls, where the anatomical structure of the brains could be visualized and analyzed. Based on these endocasts, the researchers could measure brain volume and infer key aspects of cerebral organization from impressions of the brain’s structure.
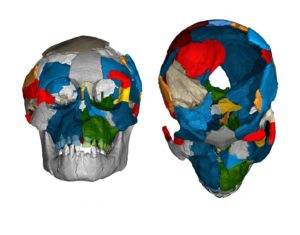
A key difference between apes and humans involves the organization of the brain’s parietal lobe–important in the integration and processing of sensory information–and occipital lobe in the visual center at the rear of the brain. The exceptionally preserved endocast of “Selam,” a skull and associated skeleton of an Australopithecus afarensis infant found at Dikika in 2000, has an unambiguous impression of the lunate sulcus–a fissure in the occipital lobe marking the boundary of the visual area that is more prominent and located more forward in apes than in humans–in an ape-like position. The scan of the endocranial imprint of an adult A. afarensis fossil from Hadar (A.L. 162-28) reveals a previously undetected impression of the lunate sulcus, which is also in an ape-like position.
Some scientists had conjectured that human-like brain reorganization in australopiths was linked to behaviors that were more complex than those of their great ape relatives (e.g., stone-tool manufacture, mentalizing, and vocal communication). Unfortunately, the lunate sulcus typically does not reproduce well on endocasts, so there was unresolved controversy about its position in Australopithecus.
“A highlight of our work is how cutting-edge technology can clear up long-standing debates about these three million-year-old fossils,” notes coauthor Kimbel. “Our ability to ‘peer’ into the hidden details of bone and tooth structure with CT scans has truly revolutionized the science of our origins.”
A comparison of infant and adult endocranial volumes also indicates more human-like protracted brain growth in Australopithecus afarensis, likely critical for the evolution of a long period of childhood learning in hominins.
In infants, CT scans of the dentition make it possible to determine an individual’s age at death by counting dental growth lines. Similar to the growth rings of a tree, virtual sections of a tooth reveal incremental growth lines reflecting the body’s internal rhythm. Studying the fossilized teeth of the Dikika infant, the team’s dental experts calculated an age at death of 2.4 years.
The pace of dental development of the Dikika infant was broadly comparable to that of chimpanzees and therefore faster than in modern humans. But given that the brains of Australopithecus afarensis adults were roughly 20 percent larger than those of chimpanzees, the Dikika child’s small endocranial volume suggests a prolonged period of brain development relative to chimpanzees.
“The combination of apelike brain structure and humanlike protracted brain growth in Lucy’s species was unexpected,” says Kimbel. “That finding supports the idea that human brain evolution was very much a piecemeal affair, with extended brain growth appearing before the origin of our own genus, Homo.”
Among primates, different rates of growth and maturation are associated with different infant-care strategies, suggesting that the extended period of brain growth in Australopithecus afarensis may have been linked to a long dependence on caregivers. Alternatively, slow brain growth could also primarily represent a way to spread the energetic requirements of dependent offspring over many years in environments where food is not always abundant. In either case, protracted brain growth in Australopithecus afarensis provided the basis for subsequent evolution of the brain and social behavior in hominins and was likely critical for the evolution of a long period of childhood learning.
_________________________

Brain imprints in fossil skulls of the species Australopithecus afarensis (famous for “Lucy” and the “Dikika child” from Ethiopia pictured here) shed new light on the evolution of brain growth and organization. The exceptionally preserved endocranial imprint of the Dikika child reveals an ape-like brain organization, and no features derived towards humans. Philipp Gunz, MPI EVA Leipzig.
_________________________
Article Source: ARIZONA STATE UNIVERSITY news release
_________________________
Advertisement





